|
The
Conserved Asparagine in the HNH Motif Serves an Important
Structural Role in Metal Finger Endonucleases
Hsinchin Huang and Hanna S. Yuan,*
Institute
of Molecular Biology, Academia Sinica, Taipei, Taiwan 11529, Republic of China
ABSTRACT
The HNH motif is a small nucleic acid
binding and cleavage module, widespread in metal finger endonucleases
in all life kingdoms. Here we studied a non-specific endonuclease,
the nuclease domain of ColE7 (N-ColE7), to decipher the role of the conserved
asparagine and histidine
residues in the HNH motif.We found, using
fluorescence resonance energy transfer (FRET) assays, that the DNA hydrolysis
activity of H545 N-ColE7 mutants was completely abolished while activities of
N560 and H573
mutants varied from 6.9% to 83.2% of the wild-type activity.
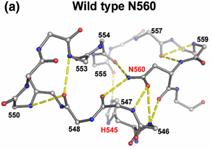
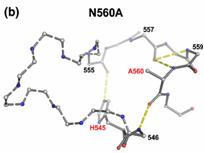
The crystal structures of three N-ColE7 mutants in complex with the inhibitor
Im7, N560A–Im7, N560D–Im7 and H573A–Im7, were determined at a
resolution of 1.9 Å to 2.2 Å. H573 is responsible for metal ion
binding in the wild-type protein, as the zinc ion is still partially
associated in the structure of H573A,suggesting that H573 plays a supportive role in metal
binding. Both N560A and
N560D contain a disordered loop in the HNH motif due to the disruption of the
hydrogen bond network surrounding the side-chain of residue 560, and as a result,
the imidazole ring of the general base residue H545
is tilted slightly and the scissile phosphate is shifted, leading to the
large reductions in hydrolysis activities. These results suggest that the
highly conserved asparagine in the HNH motif, in
general, plays a
structural role in constraining the loop in the metal finger
structure and keeping the general
base histidine and scissile phosphate in the
correct position for DNA
hydrolysis.
|


