ABSTRACT
Human Tudor-SN is involved in the
degradation of hyper-edited inosine-containing microRNA precursors,thus
linking the pathways of RNA interference and editing. Tudor-SN contains
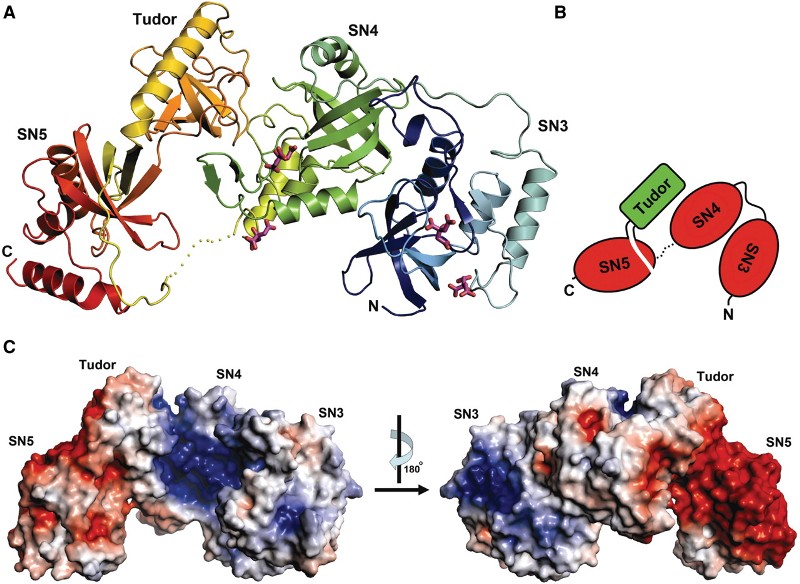
four tandem repeats of staphylococcal nuclease-like domains
(SN1–SN4) followed by a tudor and C-terminal SN domain (SN5).
Here, we showed that Tudor-SN requires tandem repeats of SN domains for
its RNA binding and cleavage activity. The crystal structure of a 64-kD
truncated form of human Tudor-SN further shows that the four domains,
SN3, SN4, tudor and SN5, assemble into a crescent-shaped structure. A
concave basic surface formed jointly by SN3 and SN4 domains is likely
involved in RNA binding, where citrate ions are bound at the putative
RNase active sites. Additional modeling studies provide a structural
basis for Tudor-SN’s preference in cleaving RNA containing multiple IU wobble-paired sequences. Collectively,
these results suggest that tandem repeats of SN domains in Tudor-SN
function as a clamp to capture RNA substrates.